| Authors | Zhen James Wang, Nicolas Daniel Spiccia, Christopher James Gartshore, Jayamini Illesinghe, William Roy Jackson and Andrea Jane Robinson |
| Published | Synthesis |
| Graphical abstract |
|
| Abstract |
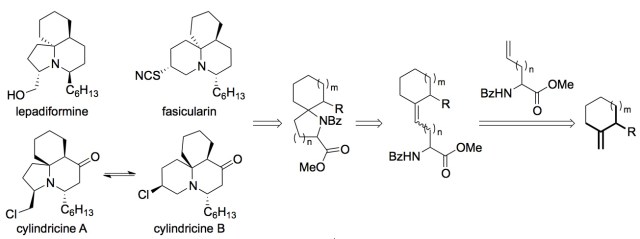
The 1-azaspirocyclic ring system can be found in a number of bioactive natural product families. Lepadiformine, cephalotaxine and cylindricine A all share a common spirocyclic pyrrolidine core, and fasicularin, halichlorine, histrionicotoxin and cylindricine B possess a homologous spirocyclic piperidine centre. This paper describes a concise synthesis of spiropyrrolidines and spiropiperidines which employs a ruthenium–alkylidene-catalysed cross-metathesis reaction of enantiopure N-protected allylglycine with methylenecycloalkanes. The resultant alkene intermediates can then undergo a tandem acid-catalysed cyclisation to form spiropyrrolidines. Ring expansion of the spiropyrrolidine system, via an aziridinium intermediate, grants access to the homologous spiropiperidine ring system with excellent stereo-retention. |
| Citation | Z. J. Wang, N. D. Spiccia, C. J. Gartshore, J. Illesinghe, W. R. Jackson, A. J. Robinson, Synthesis, 2013, 45(22), 3118-3124. |
| Article | |
| Doi | DOI: 10.1055/s-0033-1338527 |
| Related |
-
Recent Posts
- Minimizing Mitogenic Potency of Insulin Analogues Through Modification of a Disulfide Bond
- Toward the stereoselective synthesis of zaragozic acid framework: A desilylation-aldol reaction approach
- [18 F]Ethenesulfonyl Fluoride as a Practical Radiofluoride Relay Reagent
- Bioinspired carrier-free peptide conjugated BF2-oxasmaragdyrin dye-based nano self-assemblies: a photostable NIR cancer theragnostic agent
- Design, Development, In Vitro and Preliminary In Vivo Evaluation of a Novel Photo-Angioplasty Device: Lumi-Solve
- BF2-Oxasmaragdyrin Nanoparticles: A Non-toxic, Photostable, Enhanced Non-radiative Decay-Assisted Efficient Photothermal Cancer Theragnostic Agent
- Synthesis and Studies of Glucosamine Conjugated BF2 -Oxasmaragdyrin
- Cell-Penetrating Peptide-Conjugated BF 2 -Oxasmaragdyrins as NIRF Imaging and Photothermal Agents”
Archives
Categories
Meta