| Authors |
Ellen C. Gleeson, Zhen J. Wang, W. Roy Jackson, and Andrea J. Robinson |
| Published | Journal of Organic Chemistry |
| Graphical abstract |
|
| Abstract |
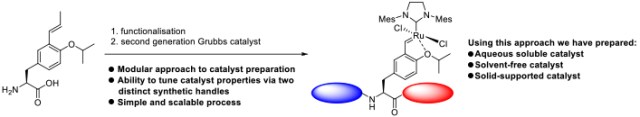
A simple and generic approach to access a new family of Ru−alkylidene olefin metathesis catalysts with specialized properties is reported. This strategy utilizes a late stage, utilitarian Hoveyda-type ligand derived from tyrosine, which can be accessed via a multigram-scale synthesis. Further functionalization allows the catalyst properties to be tuned, giving access to modified second-generation Hoveyda−Grubbs-type catalysts. This divergent synthetic approach can be used to access solid-supported catalysts and catalysts that function under solvent-free and aqueous conditions. |
| Citation |
Ellen C. Gleeson, Zhen J. Wang, W. Roy Jackson, and Andrea J. Robinson, J. Org. Chem., 2015, 80(14), 7205–7211 |
| Article | |
| Doi | https://doi.org/10.1021/acs.joc.5b01091 |
| Subjects | Ligands, Precursors, Catalysts, Olefin metathesis, Metathesis |
-
Recent Posts
- Minimizing Mitogenic Potency of Insulin Analogues Through Modification of a Disulfide Bond
- Toward the stereoselective synthesis of zaragozic acid framework: A desilylation-aldol reaction approach
- [18 F]Ethenesulfonyl Fluoride as a Practical Radiofluoride Relay Reagent
- Bioinspired carrier-free peptide conjugated BF2-oxasmaragdyrin dye-based nano self-assemblies: a photostable NIR cancer theragnostic agent
- Design, Development, In Vitro and Preliminary In Vivo Evaluation of a Novel Photo-Angioplasty Device: Lumi-Solve
- BF2-Oxasmaragdyrin Nanoparticles: A Non-toxic, Photostable, Enhanced Non-radiative Decay-Assisted Efficient Photothermal Cancer Theragnostic Agent
- Synthesis and Studies of Glucosamine Conjugated BF2 -Oxasmaragdyrin
- Cell-Penetrating Peptide-Conjugated BF 2 -Oxasmaragdyrins as NIRF Imaging and Photothermal Agents”
Archives
Categories
Meta